Meet Lab4Chemistry’s new tool: The color Inspector
By Lab4U Team
Our perception of the world depends on what our senses can capture: shapes, textures, sounds, aromas, flavors, and … colors! Colors are perceived by the retina, a sector of the eyeball with the presence of two types of photosensitive cells. Rods are capable of perceiving silhouettes, shapes, and shadows, while cones are responsible for color vision, perceiving wavelengths of greens, reds, and blues. But what are wavelengths?
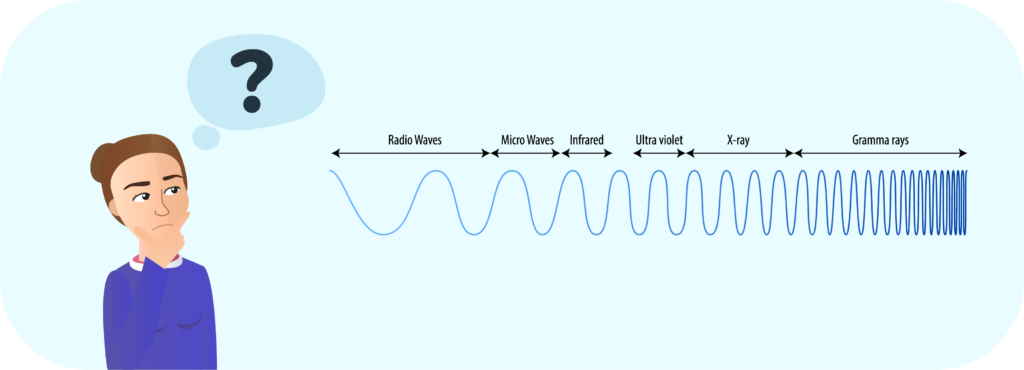
When electrons jump from a high-energy level to a low-energy level within atoms, the photons released travel at almost 300,000 km/s. Each of these photons generates an electromagnetic field through which waves of different lengths travel. Some of these waves are even smaller than an atom, such as ultraviolet radiation, and others up to kilometers in length, such as radio waves. Our eye is capable of perceiving a single section of this electromagnetic spectrum between the wavelengths of violet and those corresponding to red.
Thus, white light is considered a mixture of all the colors or wavelengths of the visible spectrum. When light falls on the surface of an object, this object is able to absorb or reflect some or all of its lengths. If all the lengths are absorbed, a black object will be observed, while if all are reflected it will appear white. Therefore, if we see an object of green color, it means that it is absorbing all the lengths, except the green one, which is being reflected. This wave reaches our retina and is captured by the green cone, which transforms this energy into a nerve impulse that travels to our brain and is interpreted in the cortex of the occipital lobe.
Why don’t we see all the objects of the same color?

As mentioned above, the color we perceive depends on the wavelength that we receive on our retina. However, this depends on the chromophores present in an object. Chromophores are sectors of a molecule that are sensitive to light and therefore responsible for its color. They can also be defined as a substance that has electrons capable of absorbing energy and becoming excited at different wavelengths. In biochemistry, it is recognized that the basic structure of a chromophore is a pyrrole group which can be presented as open-chain non-metallic pyrroles (such as carotenoids and anthocyanins) or ring pyrroles (porphyrin) with a metal ion in between, such as hemoglobin (with an iron cation) or chlorophyll (with a magnesium cation).
Due to the presence of chromophores in pigments, these are normally used as indicators in different types of reactions. Being sensitive to changes in the environment, such as pH, it is possible to alter the composition of these pyrroles, modifying the color they reflect. Likewise, if they gain or lose electrons (they are reduced or oxidized), a change in their coloration can also be observed, therefore, a color change in the substance will be observed.
Another mechanism for the production of colors in chemistry has to do with the incineration of metals. Each metal has a range of photon emission when its electrons are excited. This idea is used as a basis for the production of fireworks, which are synthesized systems made out of gunpowder and some salt. When the gunpowder explodes, the electrons in the metal of the salt are excited and release photons of a characteristic color, creating beautiful spectacles in the sky.
How does the study of colors affect the teaching of Chemistry?
Theoretically, there are countless chemical reactions and processes that occur on a daily basis, however, many of them require a significant level of abstraction. Johnstone (1991) states that chemical knowledge must be represented at three levels: macroscopic, microscopic, and symbolic. If we think of the states of aggregation of water, at the macroscopic level we think of ice, liquid water, and steam. At the molecular (microscopic) level, we can think about the degree of movement that the water molecules have and the intermolecular interactions that occur among them in the different states, while if we think at the symbolic level we can situate ourselves in the representation by means of an equation of the change from solid to liquid water and from this to gaseous, considering its enthalpy values.
Unfortunately, research in science didactics has shown that the teaching of chemistry has focused on the understanding of the symbolic level, which does not make sense without the representation of the previous levels.
One way to solve this situation is to intend the teaching of chemistry in the observation and description of macroscopic phenomena in order to find their microscopic meaning later, and their symbolic representation. From this point of view, analyzing the color change in a reaction could be a good approach.
Due to all the above, as a company, we have designed the Color Inspector, by Lab4Chemistry, a tool that can detect changes in the color of one or more substances and its intensity over time. Data collection begins with the selection of the number of samples and the time interval in which you want to capture a photograph. Subsequently, the capture of a sequence of photographs takes place over a period of time and, finally, the processing of the captured images. These results are graphed and presented in a data chart that you can export to a file.

With this tool you will have the possibility of proposing microscopic explanations of macroscopic phenomena, considering changes and intermediate states that can be difficult to perceive using only our eye. Likewise, you will be able to modify physicochemical variables capable of altering the course of a reaction, its speed, or equilibrium, measuring several systems simultaneously allowing them to be compared. Immerse yourself with us in the fascinating world of chemical reactions, kinetics, chemical balance and many other topics that you can delve into.